Research
-
Computational Modeling of Self-Assembled Nanoparticles for Drug and Gene Delivery
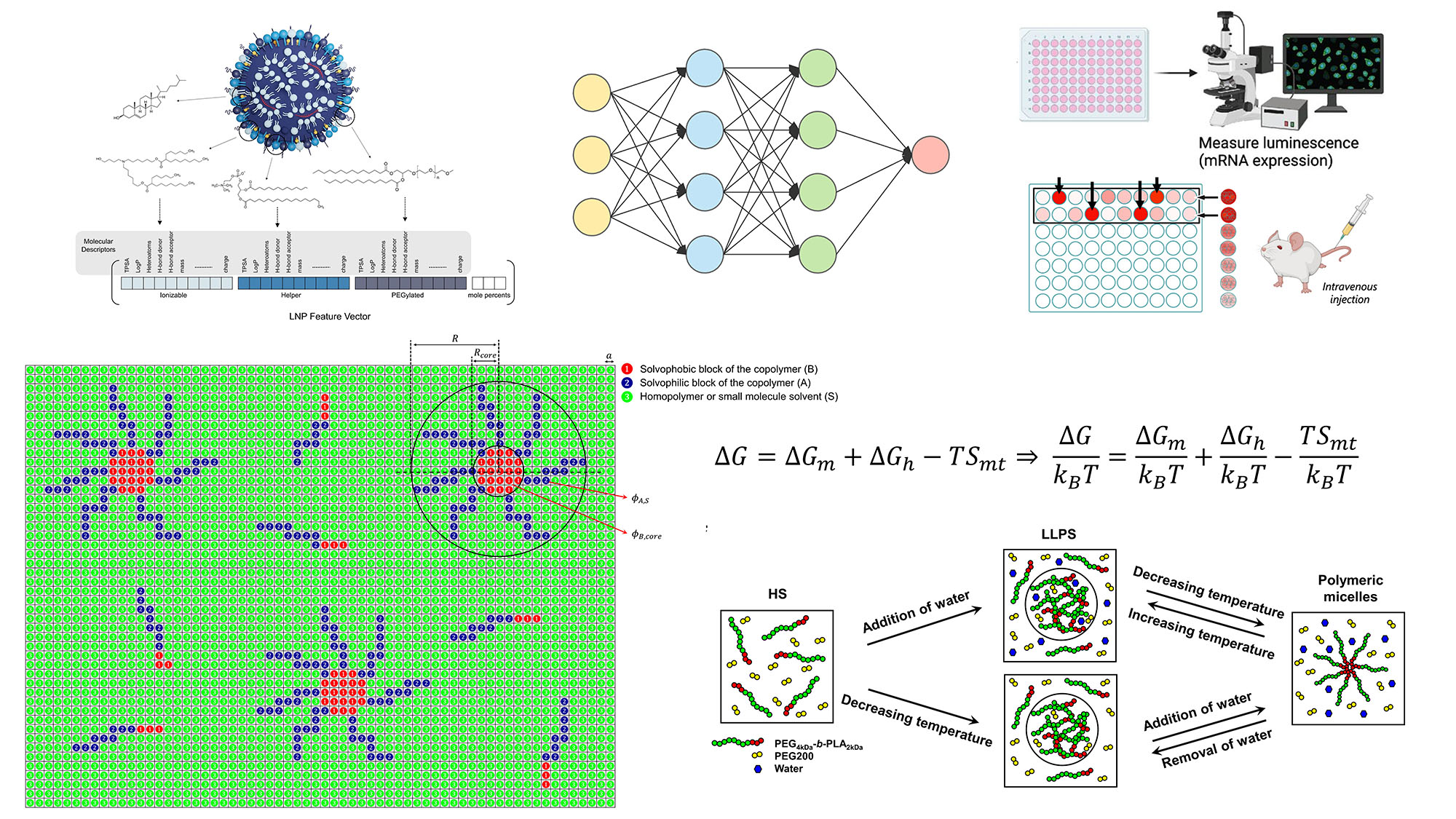
We develop computational frameworks for the predictive design of self-assembled nanoparticles, focusing on lipid nanoparticles and polymeric micelles for drug and gene delivery. Using generative AI and machine learning, we design ionizable lipids and LNP formulations and predict transfection efficiency and biodistribution, enabling strategies to reduce hepatic accumulation and enhance extrahepatic delivery. For polymeric micelles, we apply thermodynamic modeling to elucidate micelle formation mechanisms and enable predictive control over particle size and drug loading. Together, our work advances nanocarrier design from empirical optimization to rational, mechanism-driven engineering.
Description of the image: The figure schematically shows lipid nanoparticles encoded into a vector and passed through a neural network to predict biological outcomes in cells or animal models. It also depicts polymeric micelles arranged in a lattice, modeled using lattice theory and free energy equations, illustrating different phases of the system, including a homogeneous solution, liquid–liquid phase separation, and polymeric micelles.
-
Scalable Manufacturing of Nanomedicines for Solubilization, PK Control, and Precision Delivery
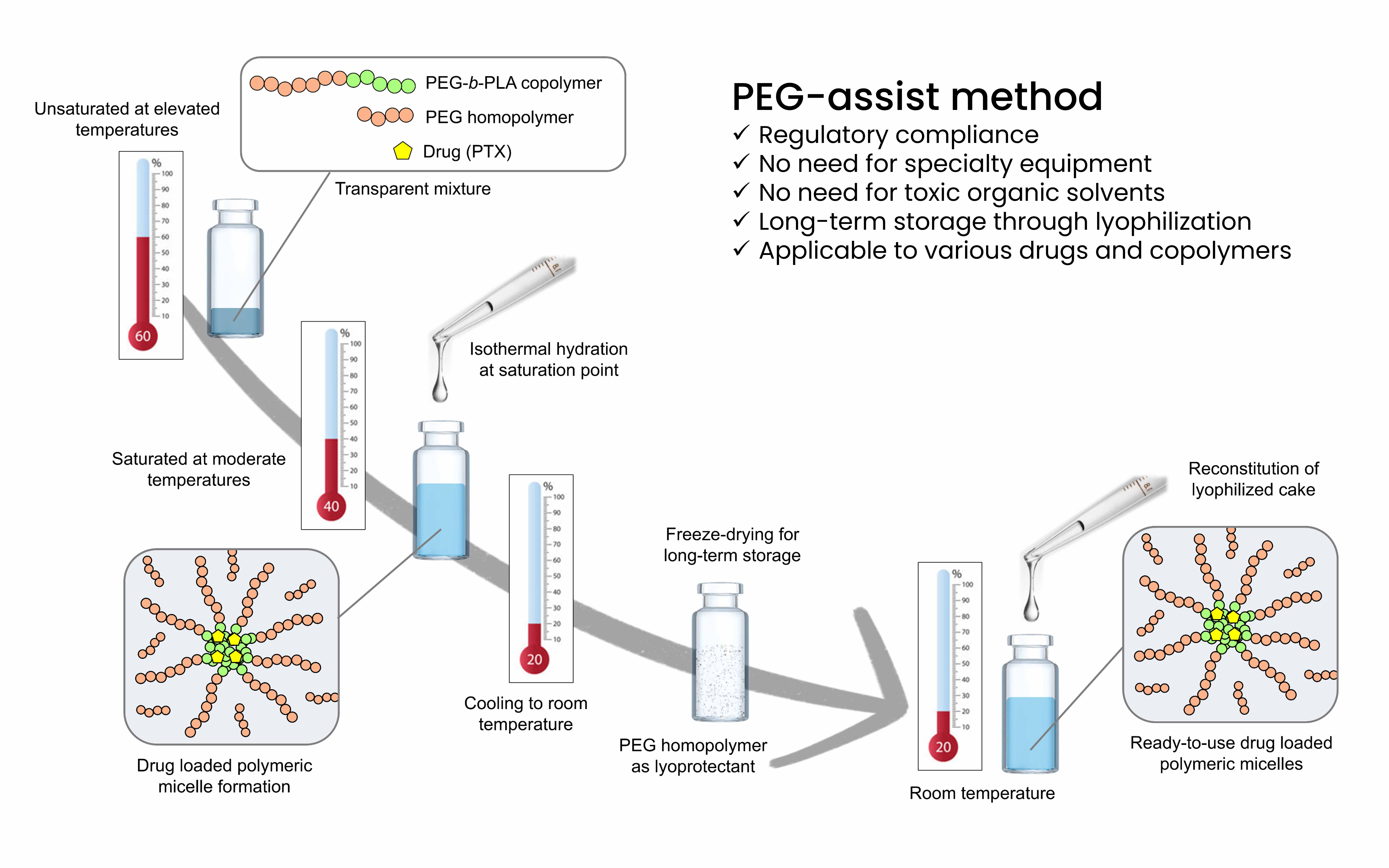 We develop scalable and reproducible manufacturing platforms for polymeric nanomedicines
to address the poor aqueous solubility and limited clinical translation of many small-molecule
therapeutics. Guided by thermodynamic and computational modeling, our work establishes
GMP–compatible processes for producing drug-loaded polymeric micelles with consistent
quality and performance. These platforms enable reliable encapsulation of diverse
hydrophobic drugs while providing precise control over micelle size, drug loading,
stability, and storage. Together, this work supports robust pharmacokinetic control
and facilitates the clinical deployment of polymeric nanomedicines for precision drug
delivery.
We develop scalable and reproducible manufacturing platforms for polymeric nanomedicines
to address the poor aqueous solubility and limited clinical translation of many small-molecule
therapeutics. Guided by thermodynamic and computational modeling, our work establishes
GMP–compatible processes for producing drug-loaded polymeric micelles with consistent
quality and performance. These platforms enable reliable encapsulation of diverse
hydrophobic drugs while providing precise control over micelle size, drug loading,
stability, and storage. Together, this work supports robust pharmacokinetic control
and facilitates the clinical deployment of polymeric nanomedicines for precision drug
delivery.Description of the image: The figure schematically shows the steps for fabricating polymeric micelles using the PEG-assisted method. This involves dissolving the copolymer, PEG, and drug at elevated temperatures, then cooling, adding water to form polymeric micelles, and freeze-drying. It also highlights the advantages of the PEG-assisted method, including regulatory compliance, no need for organic solvents or specialized equipment, long-term storage, and applicability to a range of drugs and copolymers.
-
Development of 3D Tissue Models for Drug Screening Applications
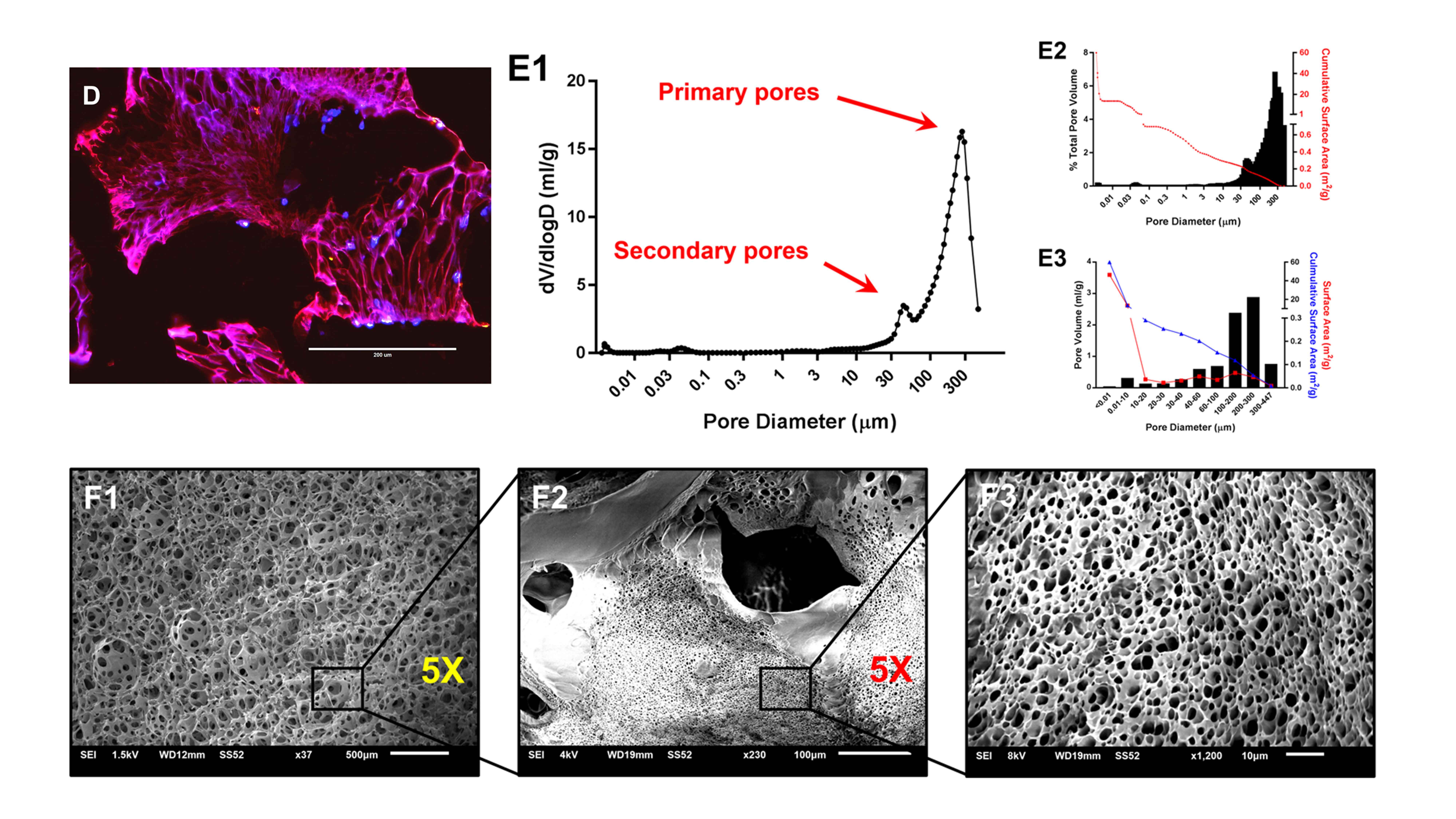 We develop three-dimensional tissue models to improve the physiological relevance
of in vitro drug screening and efficacy testing. Our work applies tissue engineering
principles to design scaffolds with tunable composition, architecture, and mechanical
properties that better recapitulate native tissue microenvironments. Using diverse
extracellular matrix components—including peptides, proteins, polysaccharides, and
synthetic polymers—we engineer spatially patterned scaffolds capable of supporting
complex multi-cellular interactions. Advanced fabrication strategies, such as microsphere
sintering, freeze-drying, and additive manufacturing, enable precise control over
scaffold geometry, porosity, and internal structure, facilitating more predictive
and translatable 3D tissue models.
We develop three-dimensional tissue models to improve the physiological relevance
of in vitro drug screening and efficacy testing. Our work applies tissue engineering
principles to design scaffolds with tunable composition, architecture, and mechanical
properties that better recapitulate native tissue microenvironments. Using diverse
extracellular matrix components—including peptides, proteins, polysaccharides, and
synthetic polymers—we engineer spatially patterned scaffolds capable of supporting
complex multi-cellular interactions. Advanced fabrication strategies, such as microsphere
sintering, freeze-drying, and additive manufacturing, enable precise control over
scaffold geometry, porosity, and internal structure, facilitating more predictive
and translatable 3D tissue models.Description of the image: The figure shows a tissue engineering scaffold with a bimodal pore size distribution, where large and small pores coexist. It includes scanning electron microscopy images of pore morphology and fluorescence images demonstrating cell attachment on the scaffold.
 David Collins Way Road Closur...
David Collins Way Road Closur...